Ni-foam supported Co(OH)F and Co–P nanoarrays for energy-efficient hydrogen production via urea electrolysis†
Abstract
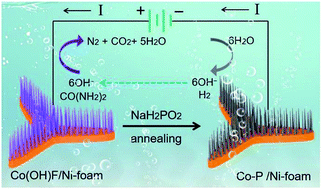
It is an urgent requirement to develop non-precious metal-based catalysts with excellent electrocatalytic activity and stability to accelerate the development of hydrogen generation via energy-efficient routes. Herein, a facile and scalable strategy was developed to synthesize both rod-like Co(OH)F and Co–P nanoarrays supported on Ni-foam, denoted as Co(OH)F/NF and Co–P/NF, respectively. Electrochemical measurements demonstrate that Co–P/NF exhibits excellent electrocatalytic performance for the hydrogen evolution reaction (HER), delivering a low overpotential of 70 mV and 43 mV at 10 mA cm−2 in alkaline and acid media, respectively. Furthermore, the as-prepared Co(OH)F/NF contributes to an outstanding oxygen evolution reaction (OER) performance with a low oxidation potential of about 1.5 V at 10 mA cm−2. In addition, the Co(OH)F/NF also can enable highly efficient urea oxidation reaction (UOR) electrocatalysis, which can be utilized to substitute OER to lower the overpotential and thus reduce electrical energy consumption during H2-production. As a proof of concept, full water-splitting measurements were performed with Co–P/NF and Co(OH)F/NF as cathode and anode, respectively, in 1 M KOH with 0.7 M urea. The Co–P/NF‖Co(OH)F/NF electrode is capable of producing a current density of 20 mA cm−2 at a cell potential of only 1.42 V, which is 230 mV less than that for the urea-free counterpart, demonstrating its potential feasibility in practical applications of energy-efficient hydrogen production.
- This article is part of the themed collection: 2019 Journal of Materials Chemistry A Most Popular Articles


 Please wait while we load your content...
Please wait while we load your content...