Controlling the 3-D morphology of Ni–Fe-based nanocatalysts for the oxygen evolution reaction†
Abstract
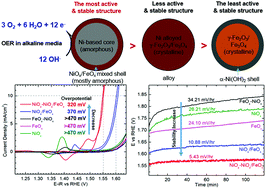
Controlling the 3-D morphology of nanocatalysts is one of the underexplored but important approaches for improving the sluggish kinetics of the oxygen evolution reaction (OER) in water electrolysis. This work reports a scalable, oil-based method based on thermal decomposition of organometallic complexes to yield highly uniform Ni–Fe-based nanocatalysts with a well-defined morphology (i.e. Ni–Fe core–shell, Ni/Fe alloy, and Fe–Ni core–shell). Transmission electron microscopy reveals their morphology and composition to be NiOx–FeOx/NiOx core-mixed shell, NiOx/FeOx alloy, and FeOx–NiOx core–shell. X-ray techniques resolve the electronic structures of the bulk and are supported by electron energy loss spectroscopy analysis of individual nanoparticles. These results suggest that the crystal structure of Ni is most likely to contain α-Ni(OH)2 and that the chemical environment of Fe is variable, depending on the morphology of the nanoparticle. The Ni diffusion from the amorphous Ni-based core to the iron oxide shell makes the NiOx–NiOx/FeOx core-mixed shell structure the most active and the most stable nanocatalyst, which outperforms the comparison NiOx/FeOx alloy nanoparticles expected to be active for the OER. This study suggests that the chemical environment of the mixed NiOx/FeOx alloy composition is important to achieve high electrocatalytic activity for the OER and that the 3-D morphology plays a key role in the optimization of the electrocatalytic activity and stability of the nanocatalyst for the OER.
- This article is part of the themed collection: National Nanotechnology Day


 Please wait while we load your content...
Please wait while we load your content...
