Bifunctional silanol-based HBD catalysts for CO2 fixation into cyclic carbonates†
Abstract
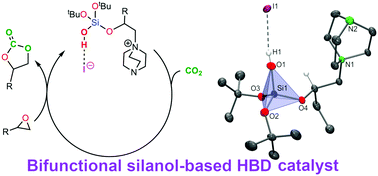
First examples of unprecedented silanol-based bifunctional HBD catalysts [(tBuO)2{(N(CH2CH2)3N)CH2CH2O}Si(OH)]+I− and (Rac)- and (R)-[(tBuO)2{(N(CH2CH2)3N)CH2(Et)CHO)}Si(OH)]+I− with tetraalkylammonium units directly incorporated into their structures were prepared from tailor-made silanols. These bifunctional silanols were used together with other mixed alkoxysilanols of general formula (tBuO)2(RO)SiOH (R = Me, Et, iPr, –CH2CH2I and –CH(Et)CH2I) in a systematic study of their catalytic properties for the preparation of cyclic carbonates using a library of epoxides and industrial-grade carbon dioxide. With 4 mol% catalyst loading in the absence of a solvent and an external nucleophile source, the bifunctional catalysts showed good to very good conversion of epoxides to the corresponding cyclic carbonates within 10 h at 70 °C and 75 psi of CO2. Furthermore, the developed synthetic approach used in the preparation of these mixed alkyl silanols via the hydrolysis of the corresponding acetoxysilyl alkoxides (tBuO)2(RO)Si(OAc) (R = organic moiety) allowed a straightforward route to the modification of the steric bulk around the silicon atom and the introduction of functional groups for further derivatization.
- This article is part of the themed collection: Celebrating recent chemical science in Mexico


 Please wait while we load your content...
Please wait while we load your content...