Multivalent ion conduction in solid polymer systems
Abstract
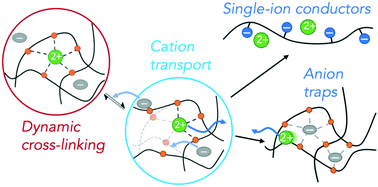
While polymer electrolytes hold the promise of improving safety and mechanical durability of electrochemical devices, most suffer from relatively low ionic conductivities especially at ambient temperature. Furthermore, much of the conductivity in polymer electrolytes stems from the mobile anions, rather than the metal cations necessary for energy storage. This combination of challenges becomes even more pronounced in the conduction of the multivalent metal ions likely to be necessary for next generation, high energy density energy storage devices where the ions are likely to have complex, multi-functional interactions with the polyelectrolyte matrix. Herein, we will review the current state of understanding of the mechanisms of multivalent ion transport through polymers and the specific challenges relative to lithium ion transport. This fundamental understanding will lead to the design of new polymer electrolytes for multivalent ion transport, including single-ion conductors and anion-trapping polymers for enhanced cation mobility.
- This article is part of the themed collection: Charge Transporting Nanostructured Polymers for Electrochemical Systems


 Please wait while we load your content...
Please wait while we load your content...
