Modelling haemoproteins: porphyrins and cyclodextrins as sources of inspiration
Abstract
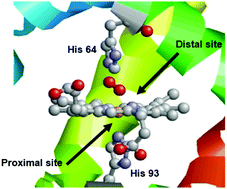
The association of hydrophobic cavities with porphyrin derivatives has been used to mimic haemoprotein structures. The most employed cavity in this field is β-cyclodextrin (β-CD), and scaffolds combining β-CDs and porphyrins are expected to inspire the combination of porphyrins and cucurbiturils in the near future. Aside from providing water solubility to various porphyrinic structures, the β-CD framework can also modulate and control the reactivity of the metal core of the porphyrin. After a general introduction of the challenges faced in the field of haemoprotein models and the binding behavior of β-CDs, this article will discuss covalent and non-covalent association of porphyrins with β-CDs. In each approach, the role of the CD differs according to the relative position of the concave CD host, either directly controlling the binding and transformation of a substrate on the metalloporphyrin or playing a dual role of controlling the water solubility and selecting the axial ligand of the metal core. The discussion will be of interest to the cucurbituril community as well as to the cavitand community, as the information provided should be useful for the design of haemoprotein mimics using cucurbiturils.
- This article is part of the themed collection: Cucurbiturils and related cavitands


 Please wait while we load your content...
Please wait while we load your content...