Pyrazolone: a powerful synthon for asymmetric diverse derivatizations
Abstract
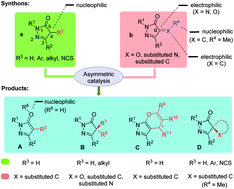
As a powerful synthon, pyrazolone has widely been exploited for the construction of various chiral pyrazoles/pyrazolones which hold great potential in the development of pharmaceutical chemistry, coordination chemistry and functional materials. Benefited from the diversity and multiple active sites of the starting materials, such as 4-nonsubstituted/4-monosubstituted pyrazolones and α,β-unsaturated pyrazolones, large numbers of strategies for the asymmetric synthesis of various pyrazoles/pyrazolones have been developed successfully in the past few years. This feature article summarizes these excellent research studies since 2015, including some efforts by our group. Based on the structural feature of the final products, this feature article is divided into four sections, including the catalytic asymmetric synthesis of 4-monosubstituted, 4-disubstituted, fused and spiro pyrazoles/pyrazolones. Schematic illustrations are given for some representative examples in each section.


 Please wait while we load your content...
Please wait while we load your content...