Binding of alkyl halides in water-soluble cavitands with urea rims†
Abstract
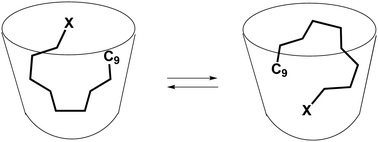
The complexation of primary alkyl halides Cn–X (n = 5, 6 and 9; X = Cl, Br and I) in a new, water-soluble cavitand is evaluated. The complexes are dynamic; the host and guest adapt their shapes to accommodate each other and the guest undergoes tumbling or yo–yo like motions in the space. Evidence for halogen bonding of the halides to the aryl surfaces of the cavitand is presented.
- This article is part of the themed collection: The halogen bond: a new avenue in recognition and self-assembly


 Please wait while we load your content...
Please wait while we load your content...