Seven-coordinate lanthanide complexes with a tripodal redox active ligand: structural, electrochemical and spectroscopic investigations†
Abstract
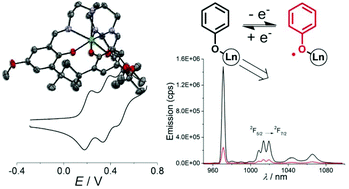
The tripodal ligand TREN-(3,5-di-tert-butylsalicylidene)3 (H3L) was synthesized and its tris(phenolato) lanthanide complexes L-Ln (Ln = NdIII, EuIII, TbIII, GdIII, ErIII, YbIII and LuIII) were prepared. The X-Ray crystal structures confirm that each metal ion resides in a similar monocapped octahedral geometry, excluding water molecules from the coordination sphere. The coordination bond distances are in agreement with the lanthanide contraction, with Ln–O bond lengths in the range 2.139–2.216 Å. The complexes show three reversible monoelectronic oxidation waves, which are assigned to the successive oxidation of the phenolate moieties to phenoxyl radicals. The L-Nd complex is the easiest to oxidize, with E11/2 = 0.11, E21/2 = 0.21 and E31/2 = 0.34 V vs. Fc+/Fc, due to the larger size of the lanthanide ion. The ΔE1/2 value (ΔE1/2 = E21/2 − E11/2) is correlated to the lanthanide radius, with values of 0.10 V for L-Nd and 0.22 V for L-Lu. The monoradical species were persistent in solution, allowing for their characterisation. All exhibit a distinct absorption band at around 445 nm due to the phenoxyl π–π* transitions. The EPR spectrum of L-Lu+ consists of a single resonance at giso = 1.999, confirming the radical nature of the oxidized product. Most of the other complexes (L-Gd, L-Er, L-Yb) show a quenching of the LnIII-based resonances upon oxidation, indicative of magnetic interactions between the metal and the radical spins. The L-Ln (L = Nd, Er, Yb) complexes exhibit a metal-based luminescence upon excitation of the ligand. A significant quenching of the luminescence was observed upon radical formation: 92%, 83% and 79% respectively for L-Nd+, L-Er+ and L-Yb+.
- This article is part of the themed collection: New Talent: Europe


 Please wait while we load your content...
Please wait while we load your content...