Metal catalyst free cyclization of 3-alkynyl substituted 2-(indol-3-yl)quinoxalines in TFA alone: a new synthesis of indolophenazines†
Abstract
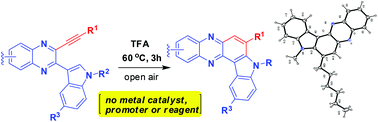
TFA alone was found to be remarkably effective for the intramolecular hydroarylation (IMHA) of alkynes when employed as a solvent in the cyclization of 3-alkynyl substituted 2-(indol-3-yl)quinoxalines. This simple and metal free cyclization method afforded a range of indolophenazines as new and potential cytotoxic agents. The use of excess TFA was found to be crucial for the success of this reaction.

 Please wait while we load your content...
Please wait while we load your content...