Hg/Pt-catalyzed conversion of bromo alkynamines/alkynols to saturated and unsaturated γ-butyrolactams/lactones via intramolecular electrophilic cyclization†
Abstract
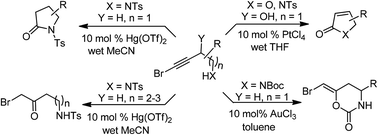
Convenient and general Hg(II)/Pt(IV) catalyzed syntheses of γ-butyrolactams and α,β-unsaturated γ-butyrolactones/lactams are described via intramolecular electrophilic cyclizations of bromoalkynes with tosylamino and hydroxyl tethers. The reaction features the use of wet solvents, the exclusion of any base and additive, mild conditions and practical yields. We also synthesised few chiral lactams through this pathway. Additionally, it is shown that the NHTs group distanced further from the homopropargylic position assists regioselective bromoalkyne hydration to yield useful α-bromoketones. Furthermore, Boc protected bromo homo propargyl amines undergo 6-endo-dig cyclization through Boc oxygen to give bromomethylene substituted oxazinones.

 Please wait while we load your content...
Please wait while we load your content...