Copper(ii) complexes with phosphorylated 1,10-phenanthrolines: from molecules to infinite supramolecular arrays†
Abstract
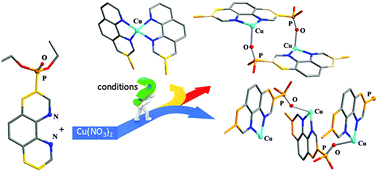
The reaction of phosphorylated 1,10-phenanthrolines 3-Pphen, 3,8-Pphen and 4,7-Pphen (3-Pphen = 3-diethoxyphosphorylphenanthroline, 3,8-Pphen = 3,8-bis(diethoxyphosphoryl)phenanthroline, 4,7-Pphen = 4,7-bis(diethoxyphosphoryl)phenanthroline) and hydrated copper(II) nitrate in a 1 : 1 ratio leads to the formation of supramolecular architectures. In the 1D coordination polymer [Cu(3-Pphen)(NO3)2]n (2) the copper atom is coordinated to only one phenanthroline ligand and the coordination sphere is completed by two oxygen atoms of nitrate anions and the oxygen atom of the phosphoryl group from the neighbouring phenanthroline ligand. Complex Cu(3,8-Pphen)(NO3)2 (3) crystallizes from dichloromethane/ether in two polymorphic forms. Form 3-I is a dimeric complex in which two phenanthroline rings are parallel and offset-shifted due to the formation of two (P)O–Cu bonds. The crystals of the polymorph 3-II are composed of polymeric chains in which the coordination sphere of the metal centres and the arrangement of the mononuclear fragments are similar to those of complex 2. Complex [Cu(4,7-Pphen)(NO3)2]2 (4) crystallizes from toluene/acetonitrile and exhibits a dimeric structure which is similar to that of 3-I.
- This article is part of the themed collection: Nitrogen Ligands

 Please wait while we load your content...
Please wait while we load your content...