Stability of solution-processed MAPbI3 and FAPbI3 layers†
Abstract
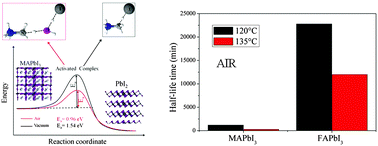
We provide a semi-empirical model based on in situ degradation measurements to predict the durability of hybrid perovskite materials under simulated thermal operation conditions. In the model, the degradation path of MAPbI3 layers is proved to follow an Arrhenius-type law. The predictive role is played by the activation energy combined with its pre-exponential factor. Our comparative study under moisture conditions with respect to vacuum and nitrogen treatments has assessed the occurrence of an intrinsic dynamic exchange of protons between the organic cations and the inorganic cage with a direct impact on the lattice stability, for which the presence of water molecules is not mandatory. This mutual interaction produces defects inside the material and volatile species, such as HI, CH3NH2 or MAI, with an associated experimental activation energy of 1.54 eV measured under vacuum conditions in dark. This value is comparable to that calculated by the density functional theory for defect generation in MAPbI3. In air, the action of water molecules reduces the activation energy for proton exchanges in dark to 0.96 eV. As an alternative solution to increase the material stability, we demonstrate that the substitution of methylammonium (MA+) with the formamidinium (FA+) cations inside the inorganic cage gives greater robustness to the overall lattice and extends the material durability due to a different interaction between the organic molecules and the inorganic cage. This definitely supports the use of FAPbI3 in applications, provided its structure can be stabilized in the dark phase at room temperature.
- This article is part of the themed collection: 2016 most accessed PCCP articles

 Please wait while we load your content...
Please wait while we load your content...