Catalytic C–F bond activation of geminal difluorocyclopropanes by nickel(i) complexes via a radical mechanism†
Abstract
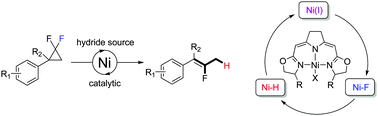
Nickel(II) fluorido complexes bearing NNN-pincer ligands were found to be catalysts in the hydrodefluorination of geminal difluorocyclopropanes which undergo ring-opening to form the corresponding monofluoroalkenes in good yield and high Z-selectivities. Evidence for a radical based mechanism involving nickel(I) and nickel hydrido complexes as key intermediates was obtained in the corresponding stoichiometric reactions.

 Please wait while we load your content...
Please wait while we load your content...