Indolizine synthesis via Cu-catalyzed cyclization of 2-(2-enynyl)pyridines with nucleophiles†
Abstract
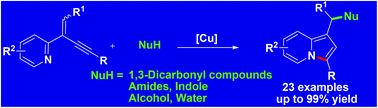
Efficient and atom-economical access to indolizines has been developed by a Cu-catalyzed cyclization of 2-(2-enynyl)pyridines with various nucleophiles through simultaneous formation of a C–N bond and a remote carbon–nucleophile bond. 1,3-Dicarbonyl compounds, indoles, amides, alcohol, and even water, were used as nucleophiles in this reaction. Good to excellent yields of the corresponding indolizines were obtained under mild reaction conditions.
- This article is part of the themed collection: HOT articles in Organic Chemistry Frontiers in 2015

 Please wait while we load your content...
Please wait while we load your content...