Expedient Ni(OTf)2/visible light photoredox-catalyzed annulation of donor–acceptor cyclopropanes with cyclic secondary amines†
Abstract
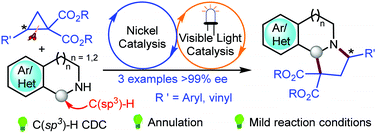
The annulative coupling of donor–acceptor cyclopropanes with cyclic secondary amines is reported using the combination of Ni(OTf)2 and visible light assisted eosin Y catalysis for tandem C–N and C–C bond formation. The reaction sequence provides a potential synthetic tool for the construction of pyrrolotetrahydroisoquinolines.


 Please wait while we load your content...
Please wait while we load your content...