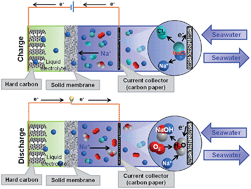
In this work, the design of a new metal-free hybrid seawater fuel cell consisting of a flowing seawater cathode and a hard carbon anode was proposed. The electrochemical performance of the cell was investigated with two different electrolytes, i.e., 1 M NaClO4 in ethylene carbonate (EC)/propylene carbonate (PC), and 1 M NaCF3SO3 in tetraethylene glycol dimethyl ether (TEGDME). The TEGDME-based electrolyte showed a good cycle performance for 100 cycles, whereas EC/PC showed poor cycle stability after 30 cycles. Our results showed that a low conducting solid-electrolyte interphase (SEI) was formed with a thick layer, and the PVdF binder was degraded during the redox reaction when the EC/PC-based electrolyte was used. In contrast, the TEGDME-based electrolyte induced the formation of a more efficient SEI layer without degradation of the binder.