Hydrothermal growth and characterization of shape-controlled NH4V3O8
Abstract
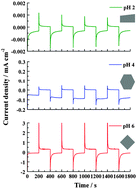
The growth of ammonium trivanadate coatings was carried out on fluorine doped tin dioxide glass substrates by the hydrothermal method using an aqueous solution of ammonium metavanadate and hydrochloric acid to adjust the pH. Among the various pH values, the coating grown at pH 6 and 95 °C after 2 h exhibited the highest electrochemical activity and the fastest time response. In addition, the kinetics of lithium ions for the oxides prepared at pH 2 was found to be affected by the deposition period. These findings are discussed in terms of possible variations in crystals' shape, crystallinity and thickness occurring at the different pH and deposition periods studied. The importance of achieving crystalline materials exhibiting the best electrochemical performance is presented.
- This article is part of the themed collection: Advanced Complex Inorganic Nanomaterials

 Please wait while we load your content...
Please wait while we load your content...