Head-to-tail photodimerization of a thiophene in a co-crystal and a rare adipic acid dimer in the presence of a heterosynthon†
Abstract
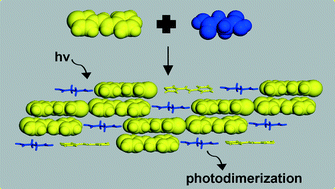
A saturated dicarboxylic acid is employed to achieve a photoreactive hydrogen-bonded assembly based on a thiophene that reacts to generate a head-to-tail photoproduct. A co-crystal that exhibits both a homo- and heterosynthon in the same co-crystal based on adipic acid and the thiophene is also presented.
- This article is part of the themed collection: Functional Co-crystals

 Please wait while we load your content...
Please wait while we load your content...