Assembly of organometallic networks with multinuclear silver(i)–ethynediide supramolecular synthon, trifluoroacetate and ligands derived from isomeric dicyanobenzenes†
Abstract
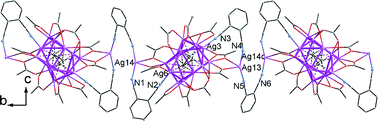
Six new silver ethynediide complexes containing trifluoroacetate and ligands derived from isomeric dicyanobenzenes, namely 2Ag2C2·10AgCF3CO2·3L1 (L1 = 1,2-dicyanobenzene) (1), 2Ag2C2·10AgCF3CO2·AgNO3·4L2 (L2 = 1,3-dicyanobenzene) (2), Ag2C2·2AgCF3CO2·2AgL3H·H2O (L3H2 = benzene-1,3-dicarboxylic acid) (3), Ag2C2·2AgCF3CO2·AgL3H·AgL4 (L4H = 3-cyanobenzenecarboxylic acid) (4), Ag2C2·4AgCF3CO2·2AgL5·4H2O (L5H = 4-cyanobenzenecarboxylic acid) (5) and 2Ag2C2·6AgCF3CO2·4AgL5·Ag2L6 (L6H2 = benzene-1,4-dicarboxylic acid) (6), have been synthesized under hydrothermal conditions and characterized using single crystal X-ray analysis. The Ln (n = 1–6) ligands incorporated into the silver ethynediide reaction system were generated in situ from hydrolysis of the o-, m- or p-isomers of dicyanobenzene. In the crystal structures of compounds 1–6, there are various types of silver(I) cages each enclosing a C22− dianion, which can be regarded as a multinuclear metal–ligand supramolecular synthon and symbolized as C2@Agn (n = 6–8). The silver cages are fused into infinite silver chains, which are connected by dicyanobenzene derivatives to yield two- and three-dimensional coordination networks in 1 and 2–6, respectively. The coordination capacity and structure-directing effect of the Ln ligands, as well as the temperature and pH value of the reaction mixture on the crystal structure of the resulting products, were also investigated.
- This article is part of the themed collection: International Year of Crystallography Celebration: Asia-Pacific

 Please wait while we load your content...
Please wait while we load your content...