Catalytic alkene cyclization reactions for the stereoselective synthesis of complex “terpenoid-like” heterocycles†
Abstract
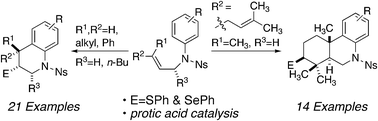
A new catalytic synthesis of densely substituted tetrahydroquinolines is described. This reaction forms up to two rings, three bonds, and three stereogenic centers with excellent stereo- and regiocontrol in a single step. Although control experiments demonstrate that the active

 Please wait while we load your content...
Please wait while we load your content...