Insights into the electrochemical degradation of phenolic lignin model compounds in a protic ionic liquid–water system†
Abstract
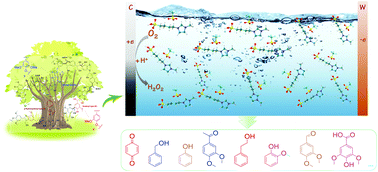
Cleavage of aryl ether (Caryl–O) bonds is crucial for the conversion and value-added utilization of lignin and its derivatives, but remains extremely challenging under mild conditions due to strong Caryl–O linkages. In this study, the Caryl–O bond breaking is achieved through electrocatalytic oxidation of four phenolic lignin model compounds with typical Caryl–O bonds, i.e., 4-ethoxyphenol (EP), 4-phenoxyphenol (PP), p-benzyloxyphenol (PBP), and 2-(2-phenylethoxy)phenol (2-PEP), in a protic ionic liquid [BSO3Hmim][OTf]–H2O electrolyte, and the electrocatalytic oxidation mechanism is also fully explored. The effects of H2O on the viscosity and conductivity of the [BSO3Hmim][OTf] ionic liquid system, as well as the solubility and diffusion coefficients of O2 and the four lignin substrates, are investigated to optimize the optimal ratio of the electrolyte system composed of [BSO3Hmim][OTf] and H2O. Electrochemical oxidation–reduction behaviors of the four lignin substrates in the [BSO3Hmim][OTf]–H2O system and the effect of O2 and N2 atmospheres on degradation are studied in detail by using cyclic voltammetry (CV) curves. Finally, by combining the analysis of degradation products with isotope labeling experiments, the C–O bond cleavage mechanism is obtained, which mainly involves direct and indirect oxidation. Specifically, under a N2 atmosphere, the substrates are oxidized directly on the RuO2-IrO2/Ti mesh electrode through Caryl–O bond splitting to form quinone and carbonium ions, while under an O2 atmosphere, apart from the direct oxidation on the electrode, indirect oxidation of the lignin substrates also occurs through in situ generated H2O2. This study may provide some insight into developing effective strategies for efficient utilization of lignin under mild conditions.
- This article is part of the themed collection: Green Chemistry 2020 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...