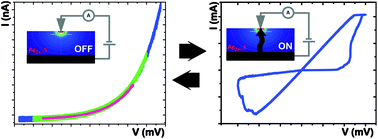
We present a quantitative analysis of the steady-state electronic transport in a resistive switching device. The device is composed of a thin film of Ag2S (solid electrolyte) contacted by a Pt nano-contact acting as ion-blocking electrode, and a large-area Ag reference electrode. When applying a bias voltage both ionic and electronic transport occurs, and depending on the polarity it causes an accumulation of ions around the nano-contact. At small applied voltages (pre-switching) we observed this as a strongly nonlinear current–voltage curve, which is modeled using the Hebb–Wagner treatment for polarization of a mixed conductor. This model correctly describes the transport of the electrons within the polarized solid electrolyte in the steady-state up until the resistance switching, covering the entire range of non-stoichiometries, and including the supersaturation range just before the deposition of elemental silver. In this way, it is a step towards a quantitative understanding of the processes that lead to resistance switching.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...