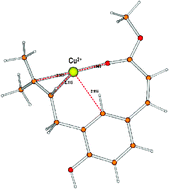
The conformational landscape of the prenylchalcone plicatin B and some of its tautomers has been investigated at the B3LYP/6-31G* level in analogy to prior studies of ours on two structurally related prenylated pterocarpans. Since the antioxidant activity of these natural compounds is supposed to be related to their copper chelation ability, several complexes with Cu(I) and Cu(II) metal cations, Cu+ and Cu2+, have been taken into account with the metal ions described by effective core potentials in the LANL2DZ valence basis set. The preferred binding sites on low-energy conformers of E and Z plicatin B have been determined and their metal ion affinity (MIA) values have been compared. Both cations give stable complexes with plicatin B, but the stability order of the metallated species at the various coordination sites strongly depends on the cation nature. In particular, for the E configuration the most stable Cu+-plicatin B ground-state structure features the metal cation bridged between the hydroxy O lone pairs and the prenyl π density, while in the most stable Cu2+ complex the cation is coordinated with the inner lone pairs of the oxygens in the methyl ester moiety bearing an anti methyl group. For the Z configuration, in contrast, the most stable Cu2+ complexes are found with the metal ions dentated between the Z ester side chain and the prenyl π density, while Cu+ in addition is close to the aromatic ring density as well. A comparison of the Cu+ and Cu2+ affinity values demonstrates however that the affinity to Cu2+ is decidedly much higher (by a factor of 3–4, depending on the arrangement type) than that to Cu+, even including the possible B3LYP overestimate of the Cu2+ binding energy with respect to BHLYP. A tentative evaluation of MIA in aqueous solution using the polarizable continuum model of the solvent shows a remarkable decrease for Cu(II).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...