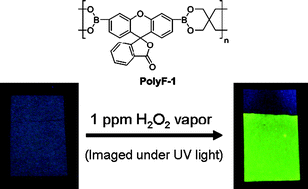
The double transesterification polymerization of 3′,6′-bis(pinacolatoboron)fluoran and pentaerythritol is reported. A model dimeric compound was synthesized to demonstrate the effectiveness of bis-diols to undergo a double transesterification, which is driven by formation of the energetically favored six-membered di-ester ring from a monomer containing a five-membered di-ester ring. This synthetic procedure provides a new route to boronate based polymers, avoiding unstable boronic acid monomers. Formation of poly-3′,6′-bis(1,3,2-dioxaborinane)fluoran, with a molecular weight of 10 000, is complete after 48 h at 50 °C. The thermodynamic stability of the six-membered boronic ester rings present in the polymer backbone also improves the stability of the polymer and its resistance to oxidation under ambient and UV light conditions. A surface detection method for the analysis of H2O2 vapor by a fluorescence turn-on response was explored. The fluorescent response results from oxidative deprotection of the boronate functionalities forming green luminescent fluorescein. Detection limits as low as 3 ppb were observed for H2O2 over an 8 h period. Detection of H2O2 in liquids can also be carried out through spot tests at concentrations as low as 1 ppm after 5 min. This new vapor-phase sensor for H2O2 provides a robust, low-cost alternative to current technology for potential applications as a self-integrating sensor for the detection of H2O2 as well as the direct monitoring of H2O2 levels in areas such as cargo shipments, chemical facilities, and pulp bleaching.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...