A simple, room temperature, solid-state synthesis route for metal oxide nanostructures†
Abstract
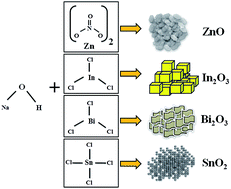
In this work, we demonstrate an extremely simple but highly effective strategy for the synthesis of various functional metal oxides (MOs) such as ZnO, In2O3, Bi2O3, and SnO2 nanoparticles with various distinct shapes at room temperature via a solid-state reaction method. The method involves only mixing and stirring of the corresponding metal salt and NaOH together in the solid phase, which yields highly crystalline metal oxides within 5–10 min of reaction time. The obtained paste can be directly doctor-bladed onto a variety of substrates for photoelectrochemical applications. The crystal structure and surface composition of the MOs are obtained by X-ray diffraction patterns, energy dispersive analysis and X-ray photoelectron spectroscopy, respectively. The surface morphology is confirmed from the scanning electron microscopy surface photo-images. The surface area and pore size distribution are studied by the N2 adsorption method. As a proof-of-concept demonstration for the application, ZnO nanoplate structures are envisaged in DSSCs as photoanodes, which enables us to obtain excellent photovoltaic properties with a power conversion efficiency of 5%. The proposed method does not require a sophisticated instrumental setup or harsh conditions, and the method is easily scalable. Hence, it can be applied for the cost-effective and large-scale production of MO nanoparticles with high crystallinity.

 Please wait while we load your content...
Please wait while we load your content...