New nonflammable tributyl phosphate based localized high concentration electrolytes for lithium metal batteries†
Abstract
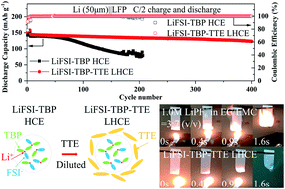
Lithium metal is one of the most promising anodes for next-generation lithium batteries. However, dendrites caused by uneven deposition can pierce the separator resulting in a safety hazard. Using nonflammable phosphates to replace conventional flammable carbonates in electrolytes is an effective solution. Here, we used tributyl phosphate (TBP), which is more thermally stable than widely studied trimethyl phosphate (TMP) and triethyl phosphate (TEP), as the main solvent for the first time to construct a fire-retardant localized high concentration electrolyte (LHCE). The results demonstrate that by adjusting the concentration of lithium bis(fluorosulfonyl)imide (LiFSI) and introducing 1,1,2,2-tetrafluoroethyl-2,2,3,3-tetrafluoropropyl ether (TTE), it eventually facilitated the formation of a LiF-rich solid electrolyte interphase (SEI) and uniform lithium deposition morphology, and a LiF-rich cathode electrolyte interphase (CEI), which effectively reduced the decomposition of the electrolyte. The viscosity and conductivity of the rationally designed LHCE were 25.5 mPa s and 257 μS cm−1, respectively. The average coulombic efficiency (CE) of the Li‖Cu is 97.3%. The Li‖LiFePO4 full cell with a limited lithium anode (50 μm) also exhibits a capacity retention of 84.6% after 400 cycles at C/2 charge and discharge. This work proposes a new solvent choice for developing high safety electrolyte systems.


 Please wait while we load your content...
Please wait while we load your content...