High-density sulfonic acid-grafted covalent organic frameworks with efficient anhydrous proton conduction†
Abstract
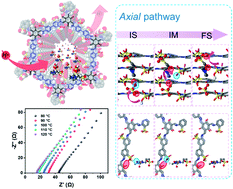
Covalent organic frameworks (COFs) have attracted much attention in the field of high-temperature anhydrous proton conduction due to their high specific surface area and ordered one-dimensional nanochannels. Herein, a high density sulfonic acid (–SO3H) functionalized proton conducting COF (TB-COF) was designed and synthesized via anchoring of sulfonic acid groups on the pore walls as a proton source. To the best of our knowledge, the synthesized TB-COF presents the highest intrinsic proton conductivity (1.52 × 10−4 S cm−1 at 120 °C) reported, due to the presence of high density –SO3H groups on the ordered 1D nanochannels. Further incorporation of a protic ionic liquid (PIL), 1-methyl-3-(3-sulfopropyl) imidazolium hydrogensulphate ([PSMIm][HSO4]), into the TB-COF greatly improved its proton conductivity to 2.21 × 10−3 S cm−1 at 120 °C. Moreover, density functional theory (DFT) calculations proved the directional migration of protons along the stacked nanochannels of the COFs with the assistance of O atoms on the keto groups. This work provides innovative structural design ideas and performance improvement methods for COFs as proton conducting materials.


 Please wait while we load your content...
Please wait while we load your content...