Design of liquid crystals with ‘de Vries-like’ properties: carbosilane-terminated 5-phenylpyrimidine mesogens suitable for chevron-free FLC formulations†
Abstract
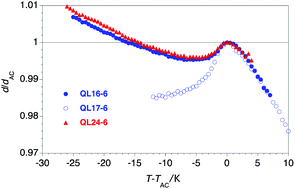
Smectic liquid crystals with ‘de Vries-like’ properties are characterized by a maximum layer contraction of ≤1% upon transition from the orthogonal SmA phase to the tilted SmC phase. In an effort to expand the library of ‘de Vries-like’ liquid crystals required for the formulation of chevron-free ferroelectric liquid crystal mixtures, we report the synthesis of a homologous series of tricarbosilane 5-phenylpyrimidine liquid crystals QL16-n using an improved synthetic route, and the characterization of their liquid crystalline and ‘de Vries-like’ properties. Measurements of orientational order parameters S2 and effective molecular lengths Leff by monodomain 2D X-ray scattering suggest that ‘de Vries-like’ behavior in series QL16-n is due to the combined effect of an increase in S2 and a decrease in bilayer interdigitation, thus causing a smectic layer expansion that compensates for the molecular tilt in the SmC phase. We also show how the optical tilt angle in the SmC phase may be optimized for SSFLC displays—without compromising ‘de Vries-like’ properties—by shortening the tricarbosilane end-group to a dicarbosilane. Two of the new materials reported herein, 5-[4-(12,12,14,14,16,16-hexamethyl-12,14,16-trisilaheptadecyloxy)phenyl]-2-hexyloxypyrimidine (QL16-6) and 2-hexyloxy-5-[4-(12,12,14,14-tetramethyl-12,14-disilapentadecyloxy)phenyl]pyrimidine (QL24-6) rank among the best ‘de Vries-like’ materials reported heretofore, with broad SmC phases and reduction factors R of 0.17 and 0.18, respectively, at a reduced temperature T − TAC = −10 K. We also show that inverting the orientation of the 5-phenylpyrimidine core in the homologous series QL17-n causes a suppression of ‘de Vries-like’ properties. These results suggest that non-covalent core–core interactions in the intercalated smectic bilayers formed by these mesogens may influence ‘de Vries-like’ behavior.

 Please wait while we load your content...
Please wait while we load your content...