Identifying key mononuclear Fe species for low-temperature methane oxidation†
Abstract
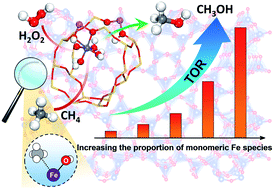
The direct functionalization of methane into platform chemicals is arguably one of the holy grails in chemistry. The actual active sites for methane activation are intensively debated. By correlating a wide variety of characterization results with catalytic performance data we have been able to identify mononuclear Fe species as the active site in the Fe/ZSM-5 zeolites for the mild oxidation of methane with H2O2 at 50 °C. The 0.1% Fe/ZSM-5 catalyst with dominant mononuclear Fe species possess an excellent turnover rate (TOR) of 66 molMeOH molFe−1 h−1, approximately 4 times higher compared to the state-of-the-art dimer-containing Fe/ZSM-5 catalysts. Based on a series of advanced in situ spectroscopic studies and 1H- and 13C- nuclear magnetic resonance (NMR), we found that methane activation initially proceeds on the Fe site of mononuclear Fe species. With the aid of adjacent Brønsted acid sites (BAS), methane can be first oxidized to CH3OOH and CH3OH, and then subsequently converted into HOCH2OOH and consecutively into HCOOH. These findings will facilitate the search towards new metal-zeolite combinations for the activation of C–H bonds in various hydrocarbons, for light alkanes and beyond.
- This article is part of the themed collection: 2021 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...