Controlling ultralong room temperature phosphorescence in organic compounds with sulfur oxidation state†
Abstract
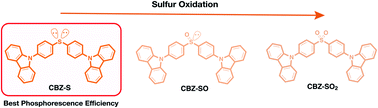
Sulfur oxidation state is used to tune organic room temperature phosphorescence (RTP) of symmetric sulfur-bridged carbazole dimers. The sulfide-bridged compound exhibits a factor of 3 enhancement of the phosphorescence efficiency, compared to the sulfoxide and sulfone-bridged analogs, despite sulfone bridges being commonly used in RTP materials. In order to investigate the origin of this enhancement, temperature dependent spectroscopy measurements and theoretical calculations are used. The RTP lifetimes are similar due to similar crystal packing modes. Computational studies reveal that the lone pairs on the sulfur atom have a profound impact on enhancing intersystem crossing rate through orbital mixing and screening, which we hypothesize is the dominant factor responsible for increasing the phosphorescence efficiency. The ability to tune the electronic state without altering crystal packing modes allows the isolation of these effects. This work provides a new perspective on the design principles of organic phosphorescent materials, going beyond the rules established for conjugated ketone/sulfone-based organic molecules.


 Please wait while we load your content...
Please wait while we load your content...