Completing the triad: synthesis and full characterization of homoleptic and heteroleptic carbonyl and nitrosyl complexes of the group VI metals†‡
Abstract
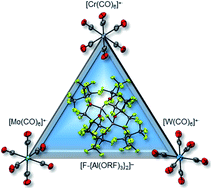
Oxidation of M(CO)6 (M = Cr, Mo, W) with the synergistic oxidative system Ag[WCA]/0.5 I2 yields the fully characterized metalloradical salts [M(CO)6]+˙[WCA]− (weakly coordinating anion WCA = [F-{Al(ORF)3}2]−, RF = C(CF3)3). The new metalloradical cations with M = Mo and W showcase a similar structural fluxionality as the previously reported [Cr(CO)6]+˙. Their reactivity increases from M = Cr < Mo < W and their syntheses allow for in-depth insights into the properties of the group 6 carbonyl triad. Furthermore, the reaction of NO+[WCA]− with neutral carbonyl complexes M(CO)6 gives access to the heteroleptic carbonyl/nitrosyl cations [M(CO)5(NO)]+ as salts of the WCA [Al(ORF)4]−, the first complete transition metal triad of their kind.
- This article is part of the themed collection: 2020 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...