Pore-size dominated electrochemical properties of covalent triazine frameworks as anode materials for K-ion batteries†
Abstract
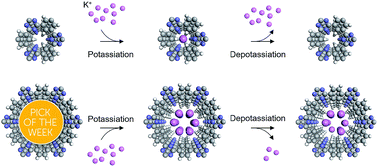
Two homologous covalent triazine frameworks (CTFs) have been developed for the first time as anode materials for high performance K-ion batteries (KIBs). The two-dimensional sheet-like structure as well as the regular channels in CTFs enable the process of intercalation/deintercalation of K-ions into/from the CTF interlayers reversibly. Particularly, a size effect of the porous structure is found to dominate the K-ion storage behavior. CTF-0 with a smaller pore size displays a higher K-ion storage capacity than CTF-1. Molecular simulations reveal the operation mechanism, showing that the depotassiation process in CTF-0 is exothermic while the depotassiation in CTF-1 is endothermic, which makes the deintercalation of K-ions from CTF-0 more feasible than from CTF-1 and contributes to the higher reversible capacity of CTF-0. This work provides a promising strategy for rational design of high-performance organic anode materials by structural modulation at the molecular scale.
- This article is part of the themed collections: 2019 Chemical Science HOT Article Collection and 2019 ChemSci Pick of the Week Collection


 Please wait while we load your content...
Please wait while we load your content...