Imidazole-modified G-quadruplex DNA as metal-triggered peroxidase†
Abstract
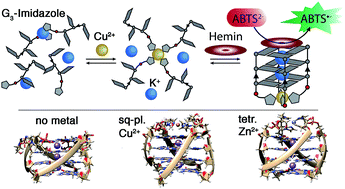
Four imidazoles, serving as metalloprotein-inspired ligands for complexing a range of transition metal cations, were incorporated into tetramolecular G-quadruplex DNA structures. Modified quadruplexes were found to complex Cu(II), Ni(II), Zn(II) and Co(II) in a 1 : 1 ratio with unprecedented strong thermal stabilizations of up to ΔT1/2 = +51 °C. Furthermore, addition of Cu(II) was found to lead to extraordinarily fast G-quadruplex association rates with kon values being ∼100 times higher compared to unmodified G-quadruplexes. This is ascribed to a template effect of Cu(II), preorganizing the four single strands via coordination, followed by rapid formation of hydrogen-bonded G-quartets. Native electrospray ionization mass spectrometry (ESI), coupled with trapped ion-mobility spectrometry (timsTOF), supports the proposed 1 : 1 G-quadruplex-metal complexes and could further disclose their ability to bind the iron–porphyrin complex hemin in a 1 : 1 stoichiometry. DNA sequence design allowed us to equip this G-quadruplex-hemin complex, known to function as a horseradish peroxidase mimic, with a metal-dependent trigger. A competitive screen of transition metals revealed a high selectivity for Cu(II), even in mixtures of several divalent metal cations. Once formed, the Cu(II)-carrying DNAzyme was shown to be preserved in the presence of EDTA, attributed to its remarkable kinetic stability. Stimuli-responsive G-quadruplexes promise application in DNAzymes with switchable activity, adaptive sensors and dynamic DNA origami constructs.


 Please wait while we load your content...
Please wait while we load your content...