The quenching effect of chitosan crosslinking on ZnO nanoparticles photocatalytic activity
Abstract
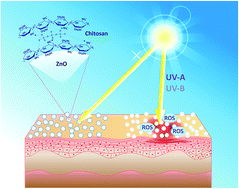
Zinc oxide (ZnO), the main component of several suntan lotions, generates highly oxidizing, cytotoxic and genotoxic reactive oxygen species (ROS) upon UV light absorption. In order to increase safety combined with ZnO use as a sunscreen, its photocatalytic activity should be efficiently quenched. In our studies commercial samples of zinc oxide nanoparticles were hybridized by ionotropic gelation with a natural biopolymer, chitosan (CS). The chemical crosslinking of the polymer in the presence of ZnO nanoparticles was performed. Significantly, in contrast to several CS–ZnO hybrid materials described in the literature, the obtained composites maintained the UV light absorption ability, while the photocatalytic activity towards chemical and biological substrates was totally quenched. Furthermore, a complete lack of photoelectrochemical response observed for the chitosan modified semiconductors confirmed the total inhibition of photoinduced interfacial electron transfer processes. Additionally, antibacterial activity against selected bacterial strains, Staphylococcus aureus and Escherichia coli was observed, although there was no cytotoxic effect against human keratinocytes. The nanocomposites resolve the problem of the risk associated with using semiconductor nanoparticles as ingredients of suntan lotions, cosmetics and dermatological formulations. The transparent polymeric coating allows the absorption of UV irradiation by ZnO particles and simultaneously blocks photogeneration of reactive radicals and oxygen species.

 Please wait while we load your content...
Please wait while we load your content...