Influence of carbonaceous species on aqueous photo-catalytic nitrogen fixation by titania†
Abstract
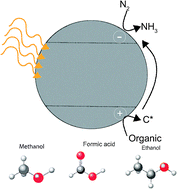
For decades, reports have suggested that photo-catalytic nitrogen fixation by titania in an aqueous environment is possible. Yet a consensus does not exist regarding how the reaction proceeds. Furthermore, the presence of an aqueous protonated solvent and the similarity between the redox potential for nitrogen and proton reduction suggest that ammonia production is unlikely. Here, we re-investigate photo-catalytic nitrogen fixation by titania in an aqueous environment through a series of photo-catalytic and electrocatalytic experiments. Photo-catalytic testing reveals that mineral phase and metal dopants play a marginal role in promoting nitrogen photofixation, with ammonia production increasing when the majority phase is rutile and with iron dopants. However, the presence of a trace amount of adsorbed carbonaceous species increased the rate of ammonia production by two times that observed without adsorbed carbon based species. This suggests that carbon species play a potential larger role in mediating the nitrogen fixation process over mineral phase and metal dopants. We also demonstrate an experimental approach aimed to detect low-level ammonia production from photo-catalysts using rotating ring disk electrode experiments conducted with and without illumination. Consistent with the photocatalysis, ammonia is only discernible at the ring with rutile phase titania, but not with mixed-phase titania. Rotating ring disk electrode experiments may also provide a new avenue to attain a higher degree of precision in detecting ammonia at low levels.
- This article is part of the themed collection: Artificial photosynthesis


 Please wait while we load your content...
Please wait while we load your content...