DFT calculation of oxygen adsorption on platinum nanoparticles: coverage and size effects†
Abstract
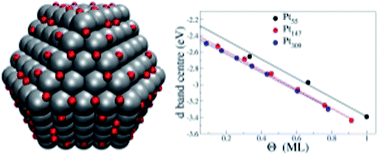
Catalysts made of Pt nanoparticles and Pt alloys are considered state-of-the-art catalysts for the anodic and cathodic reactions involved in hydrogen fuel cells. The optimal size of such nanoparticles for each chemical reaction is an unsolved problem that depends on environmental variables, such as reactant concentration, solvent, temperature, etc. From a theoretical point of view, this problem has been tackled mainly by observing how single key adsorbates react with different nanoparticles under controlled conditions. In this work, we use large-scale DFT calculations to examine the interplay between the Pt nanoparticle size and O coverage effects. We examine single O adsorptions for three adsorption sites on cuboctahedral platinum nanoparticles with different sizes. As we grow the nanoparticle size, the binding strength decreases and we observed a quick convergence of the adsorption energies with increasing nanoparticle size, which correlates with the calculated d-band centre for (111) Pt facets on such nanoparticles. We also carried out a detailed study of the effect of oxygen coverage with varying fractions of O monolayer coverage, computing adsorption energies per O atom for Pt55, Pt147 and Pt309 nanoparticles with several O coverages. In general, an increase of O coverage led to weaker adsorption energies per O atom, and when analysing the results in terms of oxygen monolayers, this effect is more pronounced for larger nanoparticles. The O coverage dependency of the adsorption energy per O atom is analysed in terms of the O distribution for each nanoparticle size and electronic changes that the adsorbed oxygen causes to the Pt nanoparticle. In studying nanoparticle size and oxygen coverage effects simultaneously, we offer insights with DFT accuracy to help on heterogeneous catalyst design.
- This article is part of the themed collection: Designing Nanoparticle Systems for Catalysis


 Please wait while we load your content...
Please wait while we load your content...