Direct observation of the external force mediated conformational dynamics of an IHF bound Holliday junction†
Abstract
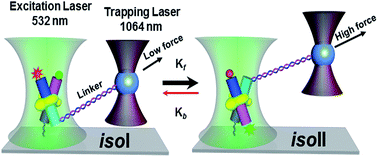
We have investigated the isomerization dynamics and plausible energy landscape of 4-way Holliday junctions (4WHJs) bound to integration host factor (IHF, a DNA binding protein), considering the effect of applied external force, by single-molecule FRET methods. A slowing down of the forward as well as the backward rates of the isomerization process of the protein bound 4WHJ has been observed under the influence of an external force, which indicates an imposed restriction on the conformational switching. This has also been reflected by an increase in rigidity, as observed from the increase in the single-molecule FRET (smFRET)-anisotropy values (0.270 ± 0.012 to 0.360 ± 0.008). The application of an external force has assisted the conformational transitions to share the unstacked open structure intermediate, with different rate-limiting steps and a huge induced variation in the energy landscape. Furthermore, the associated landscape of the 4WHJ is visualized in terms of rarely interconverting states embedded into the two isoforms by using nonlinear dynamics analysis, which shows that the chaoticity of the system increases at intermediate force (0.4 to 1.6 pN). The identification of chaos in our investigation provides useful information for a comprehensive explanation of the origin of the complex behavior of the system, which effectively helps us to perceive the dynamics of IHF bound 4WHJs under the influence of external force, and also demonstrates the applicability of nonlinear dynamics analysis in the field of biology.
- This article is part of the themed collection: Photoinduced Processes in Nucleic Acids and Proteins


 Please wait while we load your content...
Please wait while we load your content...