Reactive polycyclic aromatic hydrocarbon dimerization drives soot nucleation†
Abstract
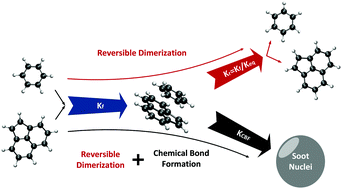
Nucleation is an important, yet poorly understood step in soot formation. Here, the importance of reactive PAH dimerization in reducing soot nucleation reversibility is investigated by simulating soot formation in a so-called “nucleation” flame (P. Desgroux et al., Combust. Flame, 2017, 184, 153–166). There, inception of soot particles is prolonged at minimal subsequent growth. With only reversible PAH dimerization, the simulated soot concentration is negligible. Accounting however for PAH chemical bond formation after physical dimerization, stabilizes dimers by covalent bonds and increases the soot concentration by four orders of magnitude, in good agreement with Laser Induced Incandescence measurements. In particular, dimers of benzene with benzene, phenylacetylene, naphthalene, toluene, acenaphthylene and cyclopentapyrene make significant contributions to the total soot concentration. The abundance of dimers with small PAHs highlights the dominant role of PAH concentration over their size and dispersion forces on dimer formation. Higher collision factors are used for irreversible dimerization models using larger PAHs because of their lower concentrations and not their larger dispersion forces leading to reduced reversibility and more stable dimers. The qualitative trend of main peaks agrees well with stochastic simulations and aerosol mass spectra measured in the above “nucleation” as well as premixed flames highlighting the abundance of PAHs with five-membered rings and substituted aliphatic chains in incipient soot. The predicted number of trimers is very low, i.e. less than 3% of the total soot nuclei formed, indicating that covalently bonded PAH dimers can be the main contributors to soot nucleation.
- This article is part of the themed collection: Bunsentagung 2018: Kinetics in the Real World


 Please wait while we load your content...
Please wait while we load your content...