Biocompatible polydopamine-like particles for the removal of heavy metals at extremely low concentrations
Abstract
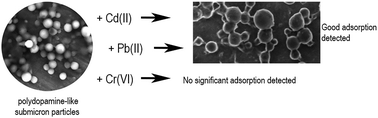
A family of catechol-based submicron particles, with sizes between 200 and 300 nm, was tested for the removal of Cd(II), Pb(II) and Cr(VI) in water. The highest adsorption capacity was obtained with catechol-based particles in the case of Pb(II), followed by Cd(II). However, the catechol particles failed to adsorb Cr(VI). Our results indicate an up to four-fold increase of the adsorption capacity of these particles compared to that of activated carbon under the same experimental conditions. To check the biocompatible character of the submicron particles, their stability was evaluated in a phosphate buffer solution (PBS) and in a cell culture medium. The results confirmed that the presence of proteins in the medium favors their stability. A bioluminescent Vibrio fischeri test and a cytotoxicity assay on the HepG2 cell line were used to determine that the catechol particles did not exhibit any substantial toxicity. The results show that these catechol-based particles can be used as an efficient biocompatible adsorbent to remove heavy metals at extremely low concentrations.

 Please wait while we load your content...
Please wait while we load your content...