Synergistic electronic modulation of Ru sites via N-doped CeO2 and Cs promotion for high efficiency H2 generation from ammonia decomposition
Abstract
Growing attention to using ammonia (NH3) as an energy carrier to produce COx-free H2 at low temperatures and supply it on demand at sites has renewed research interest. This study utilizes N-doped CeO2 material to support Ru via a solvothermal process for NH3 decomposition. The synergistic integration of N-doped CeO2 as a support and cesium (Cs) as a promoter has led to a breakthrough in catalytic activity. Advanced analysis techniques, including N2 physisorption, ICP, HAADF-STEM, CO pulse chemisorption, XRD, XPS, XAS, Raman, EPR, and temperature-programmed analysis, are employed to elucidate the beneficial properties of catalysts for the NH3 decomposition. Additionally, density functional theory (DFT) calculations provided atomic-scale insights, revealing that N-doping modulates the electronic properties of the Ru sites, thereby lowering the energy barrier for recombinative 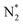 desorption and enhancing high NH3 decomposition activity at low temperatures. Ru/2N–CeO2 and Cs–Ru/2N–CeO2 exhibited superior catalytic efficiency with a H2 yield of approximately 1407 mmol min−1 gRu−1 and 2115 mmol min−1 gRu−1, respectively, and excellent stability during 100 h at 450 °C with WHSV = 30 000 mLNH3 gcat.−1 h−1. This study demonstrates that the syngenetic effects of increased oxygen vacancies (Ov) and the effective modulation of the electronic environment of Ru sites, which are optimal for enhancing recombinative N2 desorption, are vital for developing Ru-based catalysts for NH3 decomposition.
desorption and enhancing high NH3 decomposition activity at low temperatures. Ru/2N–CeO2 and Cs–Ru/2N–CeO2 exhibited superior catalytic efficiency with a H2 yield of approximately 1407 mmol min−1 gRu−1 and 2115 mmol min−1 gRu−1, respectively, and excellent stability during 100 h at 450 °C with WHSV = 30 000 mLNH3 gcat.−1 h−1. This study demonstrates that the syngenetic effects of increased oxygen vacancies (Ov) and the effective modulation of the electronic environment of Ru sites, which are optimal for enhancing recombinative N2 desorption, are vital for developing Ru-based catalysts for NH3 decomposition.



 Please wait while we load your content...
Please wait while we load your content...