Single-atom Ce synergizes icosahedral Pt2Ce nanoparticles to promote performance enhancement for zinc–air batteries
Abstract
Precious metals play an indispensable role in the development of fuel cells. Controlling the amount of noble metals added while enhancing catalytic activity and durability is of critical importance. Herein, we successfully proposed a method that utilized the synergistic effects of Pt-based nanoparticles and CeNC to enhance the oxygen reduction reaction (ORR) performance. Due to the presence of CeNC, the dispersion of Pt-based nanoparticles improved, enabling the reduction of particle size (2–3 nm). The prepared Pt2Ce alloy nanoparticles exhibited an icosahedral structure. Pt2Ce/CeNC-600 exhibited excellent performance in acidic electrolytes, with a half-wave potential (E1/2) measured at 0.81 V. When Pt2Ce/CeNC-600 was applied to zinc–air batteries (ZABs), a power density of 184 mW cm−2 was achieved during discharge. Theoretical calculations showed that electrons are transferred from CeNC to Pt2Ce in Pt2Ce/CeNC-600, indicating the presence of electron interactions and superior charge transfer ability. In addition, the energy barrier in the 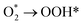 reaction was reduced because of the coexistence of Pt2Ce and CeNC, making it easier for the reactants to reach the transition state and significantly accelerating the reaction rate. This study provides a clear path for the structural control of advanced electrochemical materials.
reaction was reduced because of the coexistence of Pt2Ce and CeNC, making it easier for the reactants to reach the transition state and significantly accelerating the reaction rate. This study provides a clear path for the structural control of advanced electrochemical materials.

- This article is part of the themed collection: Journal of Materials Chemistry A HOT Papers


 Please wait while we load your content...
Please wait while we load your content...