How fluorine substituents strengthen aryl C–H bonds
Abstract
We have investigated the nature and bond dissociation energies (BDEs) of the aromatic C–H bonds in fluorinated benzenes C6R5H (each R can be H or F) using quantitative Kohn–Sham molecular orbital theory and a matching energy decomposition analysis (EDA). The C–H bond becomes stronger as the number of fluorine atoms in the benzene ring increases. This increase in the calculated BDE is additive and most pronounced for ortho-substituted C–H bonds. Our analyses of the C–H bond between 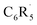 and H˙ reveal that a fluorine inductive effect is responsible for this. Fluorine polarizes the closed-shell molecular orbitals of
and H˙ reveal that a fluorine inductive effect is responsible for this. Fluorine polarizes the closed-shell molecular orbitals of 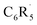 away from the carbon radical center and in this way reduces Pauli repulsion between [C˙] and the H˙ radical, leading to a stronger C–H bond. The ortho effect can be accurately modelled by a combination of Pauli repulsion (main contribution) and orbital interactions. We extend our analysis to other substituents, including ones with the opposite effect on C–H bond strength.
away from the carbon radical center and in this way reduces Pauli repulsion between [C˙] and the H˙ radical, leading to a stronger C–H bond. The ortho effect can be accurately modelled by a combination of Pauli repulsion (main contribution) and orbital interactions. We extend our analysis to other substituents, including ones with the opposite effect on C–H bond strength.

- This article is part of the themed collections: 2026 Chemical Science HOT Article Collection and #MyFirstChemSci 2026


 Please wait while we load your content...
Please wait while we load your content...