Relationship between enthalpy fluctuation and nonexponential relaxation in glass-forming liquids
Abstract
A relationship between the equilibrium fictive temperature fluctuation 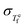 and the nonexponentiality βKWW is derived by extending the Adam–Gibbs equation within Donth's fluctuation–dissipation theorem to quantitatively account for dynamic heterogeneity in supercooled liquids. The framework is validated against three structurally, chemically, and kinetically distinct glass-forming systems, namely B2O3, Pd43Cu27Ni10P20, and PVAc, without any adjustable fitting parameters. A condition for time-temperature superposition (TTS) is further derived as a balance between the fictive temperature fluctuation rate and a configurational term composed of the configurational entropy and heat capacity difference between the glass and liquid. The present framework reveals the interplay between thermodynamic quantities, enthalpy fluctuations, and dynamic heterogeneity in glass-forming liquids.
and the nonexponentiality βKWW is derived by extending the Adam–Gibbs equation within Donth's fluctuation–dissipation theorem to quantitatively account for dynamic heterogeneity in supercooled liquids. The framework is validated against three structurally, chemically, and kinetically distinct glass-forming systems, namely B2O3, Pd43Cu27Ni10P20, and PVAc, without any adjustable fitting parameters. A condition for time-temperature superposition (TTS) is further derived as a balance between the fictive temperature fluctuation rate and a configurational term composed of the configurational entropy and heat capacity difference between the glass and liquid. The present framework reveals the interplay between thermodynamic quantities, enthalpy fluctuations, and dynamic heterogeneity in glass-forming liquids.



 Please wait while we load your content...
Please wait while we load your content...