Visible-light-driven photobiocatalytic regioselective arylation/decarboxylation cascade between chromocarb and phenylboronic acid
Abstract
In recent years, the progress of metalloenzyme catalytic technology has enabled its catalytic capacity to exceed the limitations of natural enzymes, attracting widespread attention. Inspired by natural systems, this study aims to develop a visible light-driven photoenzyme cooperative catalysis strategy that achieves a highly regioselective arylation/decarboxylation cascade between phenylboronic acid (aryl source) and chromocarb (4-oxo-4H-chromene-2-carboxylic acid) in aqueous media. A highly efficient catalytic system composed of laccase, coenzyme NADP+ and photocatalyst VB12 was established by systematic optimization and a series of flavonoid derivatives were efficiently synthesized. Mechanism studies indicated that the single-electron transfer (SET) of excited 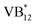 with NADP+ promotes the generation of hydroxyl radicals, which then continue to add to phenylboronic acid, leading to homolytic cleavage of the C–B bond and ultimately the formation of phenyl radicals. Meanwhile, we hypothesize that the photocatalytic cycle can activate the copper active site of laccase and promote the aggregation of phenyl radicals in the hydrophilic region near the copper protein and the completion of the oxidation decarboxylation process of chromocarb. The reaction proceeds through the SET pathway and undergoes radical generation as well as subsequent cross-coupling processes. Free radical trapping and electron paramagnetic resonance (EPR) experiments confirmed the free radical mechanism, while Stern–Volmer quenching experiments show that the enzyme enhanced the light-driven SET process. This strategy has been successfully applied to the synthesis of natural antioxidants (morin and anthocyanin) and luteolin-4-methyl ether diosgenin, providing a new biocatalysis paradigm for organic synthesis and medicinal chemistry.
with NADP+ promotes the generation of hydroxyl radicals, which then continue to add to phenylboronic acid, leading to homolytic cleavage of the C–B bond and ultimately the formation of phenyl radicals. Meanwhile, we hypothesize that the photocatalytic cycle can activate the copper active site of laccase and promote the aggregation of phenyl radicals in the hydrophilic region near the copper protein and the completion of the oxidation decarboxylation process of chromocarb. The reaction proceeds through the SET pathway and undergoes radical generation as well as subsequent cross-coupling processes. Free radical trapping and electron paramagnetic resonance (EPR) experiments confirmed the free radical mechanism, while Stern–Volmer quenching experiments show that the enzyme enhanced the light-driven SET process. This strategy has been successfully applied to the synthesis of natural antioxidants (morin and anthocyanin) and luteolin-4-methyl ether diosgenin, providing a new biocatalysis paradigm for organic synthesis and medicinal chemistry.



 Please wait while we load your content...
Please wait while we load your content...