Theoretical study on the propane dehydrogenation reaction over PtxSny intermetallic compounds
Abstract
PtxSny intermetallic compounds are widely used as catalysts in the propane dehydrogenation reaction (PDH). However, the relationships between their composition, structure and PDH performance remain unclear. In this study, the effects of PtSn catalysts (Pt3Sn, Pt1Sn1, and Pt2Sn3) and pure Pt on propane dehydrogenation with different Pt/Sn ratios are compared and studied by using density functional theory. The results show that with an increase in the Sn content, the electron density of Pt increases, thereby reducing the adsorption of propylene on the catalyst surface, and this electronic effect improves the selectivity of propylene. As the Sn content increases, the generalized coordination number 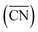 decreases, and the propylene selectivity increases, while moderate surface roughness (R) aids in the desorption of propylene. By comparing the energy barrier (Pt1Sn1(110) (1.452 eV) > Pt2Sn3(110) (1.248 eV) > Pt3Sn(111) (0.628 eV) > Pt(111) (0.535 eV)) and selectivity parameters (Ediff) (Pt2Sn3(110) (0.965 eV) > Pt1Sn1(110) (0.368 eV) > Pt3Sn(111) (0.116 eV) > Pt(111) (−0.893 eV)), it is found that the intermetallic compounds Pt3Sn and Pt2Sn3 can serve as catalysts for propane dehydrogenation. This is consistent with the experimental results. This work provides theoretical information for the rational design of high-performance Pt-based intermetallic compound catalysts for the PDH reaction.
decreases, and the propylene selectivity increases, while moderate surface roughness (R) aids in the desorption of propylene. By comparing the energy barrier (Pt1Sn1(110) (1.452 eV) > Pt2Sn3(110) (1.248 eV) > Pt3Sn(111) (0.628 eV) > Pt(111) (0.535 eV)) and selectivity parameters (Ediff) (Pt2Sn3(110) (0.965 eV) > Pt1Sn1(110) (0.368 eV) > Pt3Sn(111) (0.116 eV) > Pt(111) (−0.893 eV)), it is found that the intermetallic compounds Pt3Sn and Pt2Sn3 can serve as catalysts for propane dehydrogenation. This is consistent with the experimental results. This work provides theoretical information for the rational design of high-performance Pt-based intermetallic compound catalysts for the PDH reaction.



 Please wait while we load your content...
Please wait while we load your content...