Perlin- and Bohlmann-type effects in cyclohexylamine and 1-aminopiperidine derivatives
Abstract
Spectroscopic phenomena, such as the Perlin effect observed in 1JCH coupling constants and the Bohlmann effect in C–H stretching within infrared spectra, provide valuable insights into quantum processes like hyperconjugation and serve as tools to evaluate the conformational properties of compounds. This study investigates a Bohlmann-type effect in the N–H vibrations of piperidines and the Perlin effect in cyclohexylamine and 1-aminopiperidine derivatives. While cyclohexylamine and methyl (cyclohexylamino)acetate display N–H stretching bands typical of amines, 1-aminopiperidine and methyl [(piperidin-1-yl)amino]acetate exhibit a pronounced red shift, which remains unaffected by weak intramolecular hydrogen bonding. The Perlin effect was also observed for the set of compounds studied. Although there is a relationship of this phenomenon with 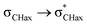 hyperconjugation, contributions from Lewis interactions are shown to be dominant, specifically the stronger steric interactions experienced by σCHeq with endocyclic σCC, while hyperconjugative interactions have an indirect effect and help modulate the magnitude of the effect. These findings underscore the utility of spectroscopic analysis in uncovering subtle yet significant interactions that govern molecular behavior, offering valuable insights for the nuanced control of molecular design.
hyperconjugation, contributions from Lewis interactions are shown to be dominant, specifically the stronger steric interactions experienced by σCHeq with endocyclic σCC, while hyperconjugative interactions have an indirect effect and help modulate the magnitude of the effect. These findings underscore the utility of spectroscopic analysis in uncovering subtle yet significant interactions that govern molecular behavior, offering valuable insights for the nuanced control of molecular design.



 Please wait while we load your content...
Please wait while we load your content...