Exploring co-crystallisation as a technique for taste-masking of nevirapine†
Abstract
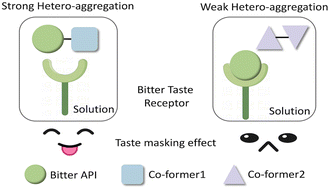
The bitter taste of pharmaceuticals majorly impacts patient adherence. Co-crystallisation has been proposed as a novel way for taste masking using sweetener-based co-formers, while other co-formers also have a positive effect. We hypothesise that the sweetness of the co-formers is not the key factor but rather the molecular aggregation between the drug and co-former in solution, i.e., the stronger the interaction, the better the taste masking effect. Here, we explore the solution aggregation between the bitter-tasting drug nevirapine and five co-formers by 1H NMR spectroscopy. The co-formers benzoic acid, salicylic acid and maleic acid show strong interaction with nevirapine, while glutaric acid and saccharin have weak and no interaction, respectively. The taste of the resulting co-crystal, as assessed by the electrical taste sensing system e-tongue, reveals that the bitterness of nevirapine has been covered with the co-crystal benzoic acid, maleic acid and glutaric acid but not saccharin or salicylic acid. From the taste results we deduct that both solution aggregation and the taste of the pure co-former play an important role in taste masking. It is likely that a large variety of co-formers can be used to cover bitter drugs and we show that the investigation of molecular aggregation in solution can help screen the co-formers before any in vitro or in vivo taste test.
- This article is part of the themed collection: RSC Pharmaceutics 2024 Hot Articles


 Please wait while we load your content...
Please wait while we load your content...