Lithiation/delithiation of silicon heavily doped with boron synthesized using the Czochralski process†
Abstract
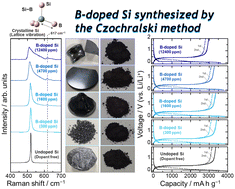
The effects of B doping and its impurity concentration (1600, 4700, and 12 400 ppm) on the electrochemical lithiation/delithiation of Si, which has the potential to replace graphite currently used as a negative electrode material in Li-ion batteries due to its huge theoretical capacity, were examined using single-crystals synthesized by the Czochralski method. XRD, Raman spectroscopic measurements, and TEM observation identified the substitution of B with Si without any impurity segregation and TOF-SIMS demonstrated that the dopant concentration was homogeneous against the depth direction. The B doping allowed Li insertion into Si at a higher voltage than the undoped Si irrespective of the B concentration, while the first delithiation capacity decreased from 3360 to 2980 mA h g−1 with the concentration increasing from 1600 to 12 400 ppm, despite the increase in electrical conductivity with increasing B doping. This result indicates the electrochemical inertness of B with Li. The heavy B doping of 12 400 ppm caused a decrease in the mechanical durability of Si, resulting in deteriorated electrode performance compared to that of the undoped Si. Even when 5 vol% fluoroethylene carbonate as a film-forming additive was added to the electrolyte and the lithiation capacity (degree of volume change) was restricted to 1000 mA h g−1, a significant improvement was not observed for the undoped Si. On the other hand, the doping at 4700 ppm provided moderate softness to Si so as not to cause the loss of the fracture toughness and its high electrical conductivity was exerted to achieve high performance with a reversible capacity of 1000 mA h g−1 at the 280th cycle.
- This article is part of the themed collection: Energy Advances: Highlight Japan & South Korea


 Please wait while we load your content...
Please wait while we load your content...