Completely non-fused ring acceptors with low non-radiative energy loss enabled by end-group modulation†
Abstract
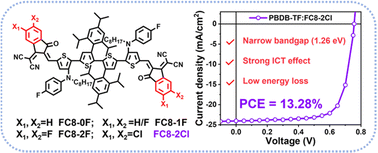
Fine tuning the end groups of non-fused ring electron acceptors (NFREAs) is one of the effective methods to reduce energy loss in organic solar cells (OSCs) and thus realize high power conversion efficiency (PCE). In this work, a series of NFREAs with fluorine substituted and chlorinated INCN end groups, FC8-0F, FC8-1F, FC8-2F and FC8-2Cl, were designed, synthesized and applied to OSCs. With the decrease of F atoms on the terminal groups, FC8-0F and FC8-1F exhibit an obvious blue-shifted absorption spectrum and a weaker intramolecular charge transfer effect than FC8-2F and FC8-2Cl. Thus, when blending the four NFREAs with the same polymer donor (PBDB-TF) to fabricate OSCs, FC8-0F- and FC8-1F-based devices show an unsatisfied PCE of 8.95% and 11.46% although they both present lower energy loss. In comparison, FC8-2Cl exhibits an enhanced intramolecular charge transfer effect and possesses an absorption range extended to about 980 nm with a narrower band-gap of 1.26 eV than FC8-2F (1.30 eV). Therefore, the FC8-2Cl-based OSC yields the highest PCE of 13.28% with a simultaneously enhanced open circuit voltage of 0.756 V, a short circuit current density of 24.03 mA cm−2 and a fill factor of 73.05% due to its relatively lower energy loss, highest extinction coefficient, fibrillar bi-continuous morphology structure and appropriate miscibility in the blends. These results demonstrate that a simple and feasible molecular design method developed by end group engineering can be used to develop low-cost non-fused acceptors with low energy loss.


 Please wait while we load your content...
Please wait while we load your content...