Highly-conductive Cu-substituted brownmillerite with emergent 3-dimensional oxygen vacancy channels†
Abstract
Lowering the oxygen content in transition-metal oxides will inevitably result in the formation of oxygen vacancies 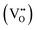 or novel crystal structures. In this work, we used Cu to substitute for Co in brownmillerite SrCoO2.5 (BM-SCO) and found that this cation-substitution strategy could effectively reduce the oxygen level. An emergent 3-dimensional
or novel crystal structures. In this work, we used Cu to substitute for Co in brownmillerite SrCoO2.5 (BM-SCO) and found that this cation-substitution strategy could effectively reduce the oxygen level. An emergent 3-dimensional 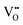 channel was formed from the parent 2-dimensional
channel was formed from the parent 2-dimensional 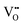 ordered BM-SCO as the Cu concentration reached 33%. The
ordered BM-SCO as the Cu concentration reached 33%. The 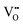 were ordered in both in-plane (IP) and out-of-plane (OOP) directions and were stabilized by forming CoO5 pyramids in addition to the existing CoO6 octahedra and CoO4 tetrahedra. Further doping with Cu would convert more and more CoO6 octahedra to CoO5 pyramids. Although the oxygen content was lowered, the electric conductivity improved by 2–3 orders of magnitude by forming an intermediate conduction band, which makes Cu-substituted BM-SCO more conductive than perovskite SCO at room temperature. Cu-substituted SCO exhibited greatly improved OER performances and boosted electrochemical activity, which is much better than that of a number of Co-based catalysts and comparable to IrO2 a noble metal oxide. Our work shows that Cu-substitution in brownmillerites is an effective route to stabilize high oxygen vacancy concentration with highly-improved conductivity. The emergent 3-dimensional oxygen vacancy channels offer a promising option to improve the catalytic performances of Co-based complex oxides.
were ordered in both in-plane (IP) and out-of-plane (OOP) directions and were stabilized by forming CoO5 pyramids in addition to the existing CoO6 octahedra and CoO4 tetrahedra. Further doping with Cu would convert more and more CoO6 octahedra to CoO5 pyramids. Although the oxygen content was lowered, the electric conductivity improved by 2–3 orders of magnitude by forming an intermediate conduction band, which makes Cu-substituted BM-SCO more conductive than perovskite SCO at room temperature. Cu-substituted SCO exhibited greatly improved OER performances and boosted electrochemical activity, which is much better than that of a number of Co-based catalysts and comparable to IrO2 a noble metal oxide. Our work shows that Cu-substitution in brownmillerites is an effective route to stabilize high oxygen vacancy concentration with highly-improved conductivity. The emergent 3-dimensional oxygen vacancy channels offer a promising option to improve the catalytic performances of Co-based complex oxides.



 Please wait while we load your content...
Please wait while we load your content...